Title: THERMOFORMABLE RIGID POLYURETHANE-POLYAMIDE FOAM
Number/Link: US2014/058005
Applicant/Assignee: BASF
Publication date: 27-2-2014
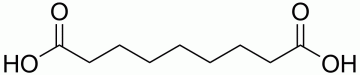
“Gist”: Rigid, open-celled and thermoformable polyurethane-polyamide foams are produced using short chain di-acids as both chain extender and blowing agent.
Why it is interesting: The reaction of a carboxylic acid group with an isocyanate group results in a mixed anhydride, which after releasing carbon dioxide forms an amide. Because the reaction is relatively sluggish a Lewis base catalyst is used, e.g. N-methylimidazole. In this case a rigid foam-forming formulation based on MDI and (pref.) polyether polyols is further reacted with short chain diacids like azelaic- or glutaric acid. The examples show foam densities of less than 30 kg/m³ without the use of water. The foams are open celled with high air flow and with a Tg of around 90°C, making them thermoformable. This type of foams is used as acoustic bonnet- en roofliners in the automotive industry.