Title: THERMOPLASTIC POLYURETHANES, PRODUCTION AND USE THEREOF
Number/Link: WO2015/155084 (German)
Applicant/Assignee: Bayer
Publication date: 15-10-2015
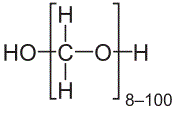
“Gist”: Paraformaldehyde-PO polyols are used to make TPU
Why it is interesting: Polyoxymethylene (POM) is a highly crystalline ‘engineering thermoplastic’ often used in blends together with TPU. In this invention POM is not blended but used to make a POM-polyether ‘block’ diol which is then used to make TPU. The diol is prepared by using paraformaldehyde as a starter which is reacted with PO (and optionally CO2) using DMC catalysis. The POM-block acts as a ‘hard block’ and use of the diol allows for an improved control of Tg, melt viscosity, hardness, chemical resistance etc. of the TPU. In the examples TPU is made from a 2000 MW parafomaldehyde/PO-CO2 block copolymer together with 4,4’MDI and BDO.