Patent Title: THERMOPLASTIC POLYURETHANE HAVING HIGH TEAR PROPAGATION STRENGTH & PROCESS FOR PRODUCING TRANSPARENT HARD THERMOPLASTIC POLYURETHANES
Number/Link: WO2018/115460 WO2018/115464
Applicant/Assignee: BASF
Publication date: 28 June 2018
“Gist”: Polyester polyols containing a “hard block” of polyethyleneterephthalate are used in the preparation of TPU
Why it is interesting: According to this invention thermoplastic polyurethanes with a high hardness at a relatively low hardblock level (<75%), or a high tear strength at relatively low Tg (<5°C) can be prepared from a polyester polyol that is a block copolymer containing 10-50% of an aromatic polyester block, the rest being aliphatic polyester. In the examples diols are prepared from PET together with adipic acid and BDO or DEG. The diols are reacted with MDI or HDI and a chain extender like BDO or HDO.
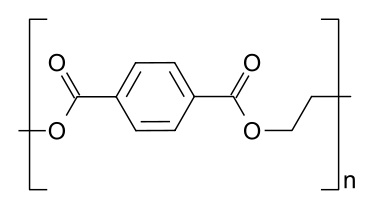
Poly(ethyeleneterephthalate)


