Patent Title: DYNAMIC UREA BONDS WITH FAST HYDROLYTIC KINETICS FOR POLYMERS
Number/Link: WO2017/155958
Applicant/Assignee: Univ. Illinois
Publication Date: 14 September 2017
“Gist”: Hindered urea bonds with fast hydrolysis kinetics are prepared from aromatic-sbstituted diamines and diisocyanates
Why it is interesting: Polyurethanes containing thermally reversible hindered urea bonds (HUBs) have been discussed before in this blog. In the current case the HUBs are prepared from aromatic (e.g aryl-) substituted amines and are said to show fast, pH-independent, hydrolysis kinetics. The HUBs can be built into linear or crosslinked polyurethanes or other polymers like polyamides, polycarbonates etc. The resulting materials are useful for medical applications like drug delivery, water-degradable packaging, self-healing materials and the like.
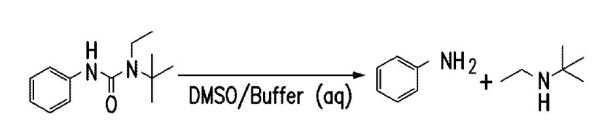
Hydrolysis reaction of phenyl-N-tetrabutyl-N-ethyl urea

