Title: POLYURETHANE HOT-MELT ADHESIVE HAVING A LOW CONTENT OF DIISOCYANATE MONOMERS AND GOOD CROSS-LINKING SPEED
Number/Link: WO2015/135833 (German)
Applicant/Assignee: Sika
Publication date: 17-09-2015
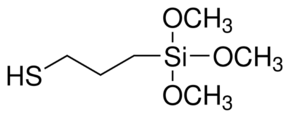
“Gist”: Use of mercaptosilane to reduce free monomeric diisocyanate
Why it is interesting: Reduction of the amount of free monomeric diisocyanate in adhesives, coatings, OCF systems etc. remains a hot topic in industrial polyurethane research, mostly because of changing legislation. For example, in the EU the amount of free MDI needs to be below 1% in order to avoid “R-40” (suspect carcinogen) labeling. A number of strategies to reduce free isocyanate have been tried in the past (and mentioned in this blog). Examples are distillation, the use of asymmetric diisocyanates and the use of monols. According to this invention the amount of free monomeric isocyanate in a hotmelt formulation can be reduced by adding a mercaptosilane like e.g. mercaptopropyltrimethoxysilane. The mercaptosilane is said to react preferentially with the monomeric isocyanate. Only a relatively small amount of mercaptosilane is used such that the main curing mechanism is still the isocyanate-water reaction.