Patent Title: BIOBASED CYCLIC CARBONATE FUNCTIONAL RESINS AND POLYURETHANE THERMOSETS THEREFROM
Number/Link: US2016/0312060
Applicant/Assignee: NDSU
Publication date: 27-10-2016
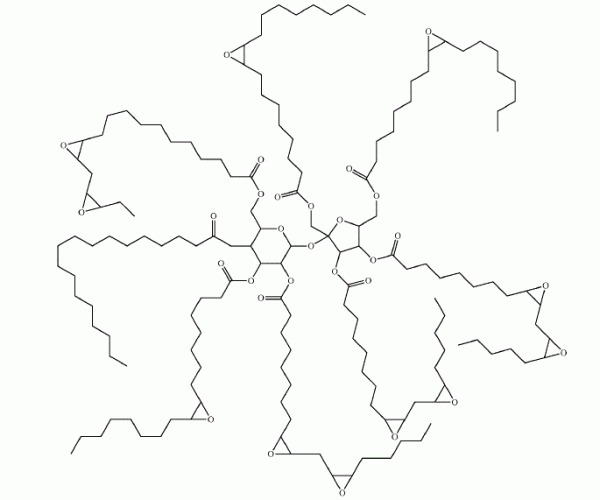
“Gist”: Epoxidized sucrose-fatty acid esters are reacted with CO2, then with polyamines
Why it is interesting: Esters of unsaturated fatty acids and sucrose have been known since the 1960s and are useful in e.g. coatings. Highly subsituted sucrose esters with an average of 7.7 fatty acid chains per molecule, a molecular weight of about 2400 and a relatively low viscosity of 300-400 mPa.s are commercially available. According to this invention, epoxidized sucrose-fatty acid esters can be further reacted with carbon dioxide resulting in a resin with a ‘pluraility’ of cyclocarbonate groups. Curing with polyamines results in highly crosslinked poly(hydroxyurethanes) useful for coatings, composites and adhesives.