Patent Title: MULTI-FUNCTIONAL CARBAMATE HAVING SOFT-SEGMENTS, POLYISOCYANATE OBTAINED VIA SUBSEQUENT NON-PHOSGENE SYNTHESIS METHODS, URETHANE PREPOLYMER AND ELASTOMERIC URETHANE HAVING SOFT-SEGMENTS DERIVED THEREFROM, AND PREPARATION METHOD THEREOF
Number/Link: US20180186918
Applicant/Assignee: Great Eastern Resins Industrial
Publication date: 5 July 2018
“Gist”: Biscarbamate with PDMS softblock backbone is heat-cracked into diisocyanates
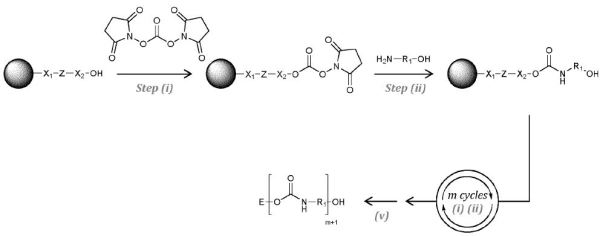
Why it is interesting: The invention relates to isocyanate-capped “softblock” siloxanyl prepared without using phosgene and polyurethanes derived therefrom. The invention is accomplished by reacting relatively high MW polydimethylsiloxanediamines with diphenylcarbonate. The resulting biscarbamate is heated under reduced pressure to produce the diisocyanate and to remove the phenol by-product. The isocyanates can be reacted with polyols and/or chain extenders to produce highly hydrophobic polyurethanes with low surface tension and good chemical resistance.
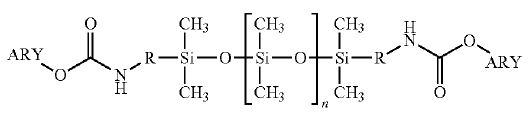
Biscarbamate with PDMS backbone